AZD9291 CAS NO.1421373-65-0
- FOB Price: USD: 100.00-100.00 /Metric Ton Get Latest Price
- Min.Order: 10 Gram
- Payment Terms: L/C,D/A,D/P,T/T,MoneyGram,Other
- Available Specifications:
Enterprise Standard(1-100)Metric Ton
- Product Details
Keywords
- AZD9291
- Osimertinib
- AZD-9291 1421373-65-0
Quick Details
- ProName: AZD9291
- CasNo: 1421373-65-0
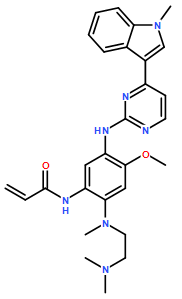
- Molecular Formula: C28H33N7O2
- Appearance: White powder
- Application: Ramelteon is a sleep agent that select...
- DeliveryTime: Immediately
- PackAge: Fluoride bottle or as required
- Port: Shanghai, Hongkong, Shenzhen
- ProductionCapacity: 30 Metric Ton/Day
- Purity: 99%
- Storage: Store in a cool and dry area, keep sea...
- Transportation: As required.
- LimitNum: 10 Gram
- Related Substances: ≤0.2%
- Residue on Ignition: 0.05%
- Heavy Metal: ≤20ppm
- Valid Period: 2 years
- Other Names: Osimertinib; mereletinib
- CAS No: 1421373-65-0
- Molecular Formula: C28H33N7O2
- Molecular Weight: 499.607
- Purity: 99%
Superiority
1.High quality
2.High purity
3.Best price
4.Best service
5.Professional manufacturer
6.Loyal and honest supplier
7.Fast response to customer within 6 hours
Product Name: AZD9291
Other Names: Osimertinib; mereletinib
CAS No: 1421373-65-0
Molecular Formula: C28H33N7O2
Molecular Weight: 499.607
Purity: 99%
Appearance: white powder
Osimertinib (previously known as mereletinib or AZD9291; trade name Tagrisso) is a third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) drug developed by AstraZeneca Pharmaceuticals – for mutated EGFR cancers.
Details
Product Name: AZD9291
Other Names: Osimertinib; mereletinib
CAS No: 1421373-65-0
Molecular Formula: C28H33N7O2
Molecular Weight: 499.607
Purity: 99%
Appearance: white powder
Osimertinib (previously known as mereletinib or AZD9291; trade name Tagrisso) is a third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) drug developed by AstraZeneca Pharmaceuticals – for mutated EGFR cancers.

| Product Name: AZD-9291 | Batch No.: BOZ/AZD/17/Jan/06 |
| CAS No.: 1421373-65-0 | Quantity: 4KG |
| Manufacturing Date: Jan., 6th, 2017 | Text Date: Jan., 7th, 2017 |
| Expiry Date: Jan., 5th, 2019 | Quality Standard: Enterprise Standard |
| TEST | SPECIFICATION | RESULT |
| Physical Characters | ||
| Appearance | White powder | Conforms |
| Identification | The retention time of text sample major peak conforms to reference standard. | Conforms |
| Impurities | ||
| single impurity |
≤0.2% |
0.18% |
| Chemical Characters | ||
| Heavy Metals | ≤20PPM | Conforms |
|
Residue on Ignition |
≤0.2% |
0.05% |
| Loss on Drying | ≤0.5% | 0.13% |
| Solvent residue |
Dioxane≤380ppm Acetone≤5000ppm Acetonitrile≤410ppm |
Not detected 482ppm 22ppm |
| Assay (by HPLC) | 98.5% min. | 99.81% |
|
Packing - Fluoride bottle or as required. Storage - Store in well closed container away from oxides and acids. |
||
Conclusion: The above analysis results comply with the specification. Customer is advised to verify the results before use.









 China (Mainland)
China (Mainland)




